- Blog
- New Page
- Download Emulador Neogeo Pc
- Yk Osiris Worth It
- Port Forwarding Software Freeware
- Planet Cnc License Crack
- Lime Torrentz Proxy
- Any Herbertos Kontakt?
- Priciples Drapping Surgical Ield
- How To Find Out Someone's Twitter Password
- Serial Number Autodesk Inventor 2013
- Uncharted 2 Game Download For Pc Highly Compressed Torrent File
- How To Calculate Engine Torque From Bore Stroke
- Jamo Sub 210 Manual
- Skeletle Flash Plugin
- Trainz Railroad Simulator 2019
- Kendrick Lamar Untitled Zip
- Eeepc Sci Raid Host Controller
- Idrac7 Licence Generator
- Human Anatomy Atlas Crack Pc
- Immunology Cv Rao Pdf
- 3d Print Alien
- Hada Crie N4000 English Manual
- Pka To Ka
In computational biology,protein pEacomputationsare utilized to estimate the gKavalues of amino acids as they can be found within protein. These calculations enhance the gTabeliefs reported for amino acids in their free of charge state, and are used often within the fields of molecular modeling, structural bioinformatics, and computational biology.
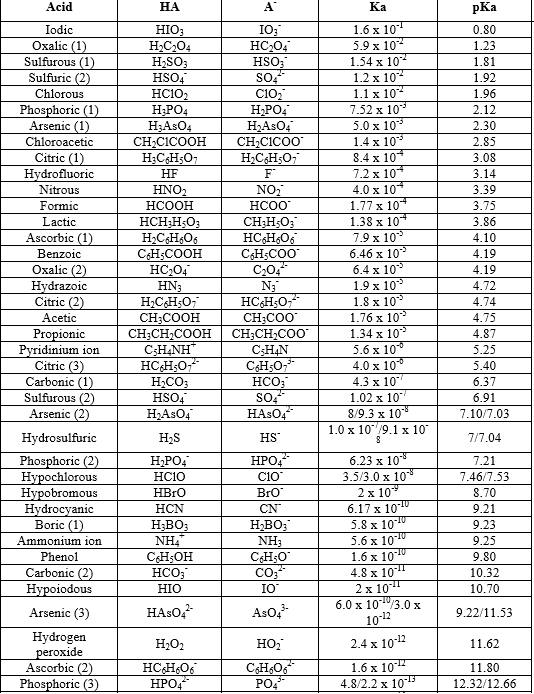
PKa Data Compiled by R. Williams page-1 pKa Values INDEX Inorganic 2 Phenazine 24 Phosphates 3 Pyridine 25 Carboxylic acids 4, 8 Pyrazine 26 Aliphatic 4, 8 Aromatic 7, 8 Quinoline 27 Phenols 9 Quinazoline 27 Alcohols and oxygen acids 10, 11 Quinoxaline 27 Amino Acids 12 Special Nitrogen Compounds 28 Peptides 13 Hydroxylamines 28. Apr 11, 2018 This organic chemistry video tutorial explains how to determine which acid is stronger using Ka and pKa values. Subscribe: https://www.youtube.com/channel/UC.
- 3pKacomputation strategies
Amino acid pKabeliefsedit
gEavalues of amino acid side chains perform an essential role in identifying the pH-dependent features of a protein. The pH-dependence of the action shown by enzymes and the pH-dependence of proteins balance, for example, are qualities that are usually identified by the pEavalues of amino acid part chains.
The pTavalues of an amino acid side string in option is usually inferred from the pKabeliefs of model substances (substances that are usually related to the aspect chains of amino acids). See Amino acid for the pKabeliefs of all amino acid aspect chains deduced in like a way. There are usually also many experimental studies that have got yielded such ideals, for instance by make use of of NMR spectroscopy.
The desk below lists the design gEaideals that are often used in a proteins gKacalculation, and contains a third column centered on proteins research.1
| Amino Acid | pTa | pTa |
|---|---|---|
| Asp (Chemical) | 3.9 | 4.00 |
| Glu (Elizabeth) | 4.3 | 4.40 |
| Arg (R) | 12.0 | 13.50 |
| Lys (E) | 10.5 | 10.40 |
| His (H) | 6.08 | 6.80 |
| Cys (G) (-SH) | 8.28 | 8.30 |
| Tyr (Y) | 10.1 | 9.60 |
| N-term | 8.00 | |
| C-term | 3.60 |
The impact of the proteins atmosphereedit
When a proteins folds, the titratable amino acids in the protein are moved from a solution-like atmosphere to an environment identified by the 3-dimensional construction of the protein. For example, in an unfolded proteins an aspartic acid usually can be in an atmosphere which reveals the titratable part chain to water. When the protein folds the aspartic acid could find itself smothered deep in the proteins interior with no publicity to solvent.
Additionally, in the folded proteins the aspartic acid will be closer to additional titratable groups in the protein and will furthermore interact with long lasting charges (age.gary the gadget guy. ions) and dipoles in the proteins.All of these effects change the pTavalue of the amino acid part chain, and pKacalculation methods generally calculate the impact of the proteins atmosphere on the design gKaworth of an amino acid part chain.2345
Usually the results of the proteins environment on the amino acid gTavalue are divided into pH-independent effects and pH-dependent results. The pH-independent effects (desolvation, relationships with permanent costs and dipoles) are usually added to the model pKaworth to give the inbuilt gKaworth. The pH-dependent results cannot become added in the same straightforward method and possess to end up being paid for for making use of Boltzmann summation, Tanford-Roxby iterations or some other methods.
The interplay of the intrinsic pKaideals of a program with the electrostatic conversation energies between titratable organizations can produce quite magnificent effects such as non-Henderson-Hasselbalch titration curves and even back-titration results.6
The picture below exhibits a theoretical program consisting of three acidic residues. One team is showing a back-titration occasion (azure group).
Coupled system consisting of three acids
gEacomputation methodsedit
Several software packages and webserver are usually available for the computation of proteins gKabeliefs. See links below or this table
Making use of the Poisson-Boltzmann equationedit
Some strategies are centered on options to the Poisson-Boltzmann equation (PBE), often referred to as FDPB-based methods (FDPBis definitely for 'finite difference Poisson-Boltzmann'). The PBE can be a adjustment of Poisson's equation that includes a description of the effect of solvent ions on the electrostatic industry around a molecule.
The H web server, the pKD webserver, MCCE, Karlsberg+, PETIT and GMCT make use of the FDPB technique to compute pTavalues of amino acid aspect chains.
FDPB-based methods calculate the shift in the pKaworth of an amino acid part string when that part chain is certainly shifted from a theoretical completely solvated condition to its place in the proteins. To carry out like a calculation, one needs theoretical strategies that can determine the effect of the proteins inside on a pEavalue, and information of the pKa beliefs of amino acid part chains in their completely solvated expresses.2345
Empirical methodsedit
A collection of empirical guidelines related the proteins construction to the gEaideals of ionizable residues possess been developed by Li, Robertson, and Jensen. These rules form the time frame for the web-accessible system called PROPKA for speedy predictions of pEavalues.A recent empirical gKaprediction plan was released by Brown KPet.al.with the on the internet server DEPTH web server
Molecular design (MD)-based methodsedit
Molecular dynamics methods of calculating gKavalues make it probable to consist of full versatility of the titrated molecule.789
Molecular design based strategies are usually much even more computationally costly, and not necessarily more accurate, methods to anticipate pKavalues than approaches centered on the Poisson-Boltzmann formula. Small conformational flexibility can furthermore be realized within a continuum electrostatics approach, e.g., for considering several amino acid sidechain rotamers. In inclusion, current frequently utilized molecular push fields do not get electronic polarizability into accounts, which could end up being an essential house in identifying protonation powers.
Identifying pEabeliefs from titration figure or free energy computationsedit
From the titration of protonatable team, one can go through the so-called pTa1⁄2which is usually similar to the pH worth where the group is certainly half-protonated. The pEa1⁄2is certainly equivalent to the Henderson-Hasselbalch pKa(gKHH
a)if the titration competition comes after the Henderson-Hasselbalch formula.10Many gKacalculation methods quietly suppose that all titration figure are usually Henderson-Hasselbalch shaped, and gEavalues in pTacomputation programs are therefore frequently identified in this method. In the general case of several interacting protonatable websites, the pEa1⁄2worth is not thermodynamically meaningful. In comparison, the Henderson-Hasselbalch pTavalue can be computed from the protonation free energy via
a)if the titration competition comes after the Henderson-Hasselbalch formula.10Many gKacalculation methods quietly suppose that all titration figure are usually Henderson-Hasselbalch shaped, and gEavalues in pTacomputation programs are therefore frequently identified in this method. In the general case of several interacting protonatable websites, the pEa1⁄2worth is not thermodynamically meaningful. In comparison, the Henderson-Hasselbalch pTavalue can be computed from the protonation free energy via
and is usually therefore in change associated to the protonation free of charge power of the site via
.
The protonation free power can in process be computed from the protonation possibility of the group ⟨x⟩(pH) which can be learn from its titration curve
Titration figure can end up being computed within a procession electrostatics approach with formally specific but more complex analytical or Monte Carlo (MC) methods, or inexact but fast approximate methods. MC methods that have been used to compute titration curves11are usually City MC12or Wang-Landau MC. Approximate methods that use a mean-field technique for computing titration curves are usually the Tanford-Roxby technique and hybrids of this technique that combine an precise statistical mechanics therapy within clusters of strongly interacting sites with a mean-field treatment of intercluster connections.1314151617
In exercise, it can be challenging to get statistically converged and precise protonation free powers from titration curves if ⟨times⟩ is certainly near to a worth of 1 or 0. In this situation, one can make use of various free of charge energy calculation methods to obtain the protonation free power11such as biased Metropolis MC,18free-energy perturbation,1920thermodynamic integration,212223the non-equilibrium work method24or the Bennett acceptance ratio method.25
Be aware that the pTHH
avalue does in general depend on the pH worth.26
avalue does in general depend on the pH worth.26
This dependence is little for weakly interacting groupings like properly solvated amino acid sidechains on the protein surface, but can be large for highly interacting groupings like those hidden in enzyme energetic sites or essential membrane proteins.272829
Work referencesedit
- ^Hass and Mulder (2015)Annu. Rev. Biophys.vol 44 pp. 53-75 doi 10.1146/annurev-biophys-083012-130351.
- ^amBashford (2004)Top Biosci.vol. 9 pp. 1082-99 doi 10.2741/1187
- ^abGunner et al. (2006)Biochim. Biophys. Actavol. 1757 (8) pp. 942-68 doi 10.1016/j.bbabio.2006.06.005
- ^acUllmann et al. (2008)Photosynth. Ers.97 vol. 112 pp. 33-55 doi 10.1007/t11120-008-9306-1
- ^anAntosiewicz et al. (2011)Mol. BioSyst.vol. 7 pp. 2923-2949 doi 10.1039/D1MB05170A
- ^A new. Onufriev, Deb.A. Case and G. M. Ullmann (2001).Biochemistry40: 3413-3419 doi 10.1021/bi002740q
- ^Donnini et al. (2011)M. Chem. Theory Comp.vol 7 pp. 1962-78 doi 10.1021/ct200061r.
- ^Wallace et al. (2011)M. Chem. Theory Comp.vol 7 pp. 2617-2629 doi 10.1021/ct200146j.
- ^Goh et al. (2012)M. Chem. Concept Comp.vol 8 pp. 36-46 doi 10.1021/ct2006314.
- ^Ullmann (2003)J. Phys. Chem. Nvol 107 pp. 1263-71 doi 10.1021/jp026454v.
- ^atUllmann et al. (2012)J. Comput. Chem.vol 33 pp. 887-900 doi 10.1002/jcc.22919
- ^Beroza et al. (1991)Proc. Natl. Acad. Sci. USAvol 88 pp. 5804-5808 doi 10.1073/pnas.88.13.5804
- ^Tanford and Roxby (1972)Biochemistryvol 11 pp. 2192-2198 doi 10.1021/bi00761a029
- ^Bashford and Karplus (1991)M. Phys. Chem.vol 95 pp. 9556-61 doi 10.1021/j100176a093
- ^Gilson (1993)Proteinvol 15 pp. 266-82 doi 10.1002/prot.340150305
- ^Antosiewicz et al. (1994)L. Mol. Biol.vol 238 pp. 415-36 doi 10.1006/jmbi.1994.1301
- ^Spassov and Bashford (1999)L. Comput. Chem.vol 20 pp. 1091-1111 doi 10.1002/(SICI)1096-987X(199908)20:11lt;1091::AID-JCC1gt;3.0.CO;2-3
- ^Beroza et al. (1995)Biophys. J.vol 68 pp. 2233-2250 doi 10.1016/S0006-3495(95)80406-6
- ^Zwanzig (1954)L. Chem. Phys.vol 22 pp. 1420-1426 doi 10.1063/1.1740409
- ^Ullmann et al. 2011L. Phys. Chem. T.vol 68 pp. 507-521 doi 10.1021/jp1093838
- ^Kirkwood (1935)L. Chem. Phys.vol 2 pp. 300-313 doi 10.1063/1.1749657
- ^Bruckner and Boresch (2011)J. Comput. Chem.vol 32 pp. 1303-1319 doi 10.1002/jcc.21713
- ^Bruckner and Boresch (2011)L. Comput. Chem.vol 32 pp. 1320-1333 doi 10.1002/jcc.21712
- ^Jarzynski (1997)Phys. Rev. Agevol pp. 2233-2250 doi 10.1103/PhysRevE.56.5018
- ^Bennett (1976)J. Comput. Phys.vol 22 pp. 245-268 doi 10.1016/0021-9991(76)90078-4
- ^Bombarda et al. (2010)L. Phys. Chem. Cvol 114 pp. 1994-2003 doi 10.1021/jp908926w.
- ^Bashford and Gerwert (1992)L. Mol. Biol.vol 224 pp. 473-86 doi 10.1016/0022-2836(92)91009-E
- ^Spassov et al. (2001)J. Mol. Biol.vol 312 pp. 203-19 doi 10.1006/jmbi.2001.4902
- ^Ullmann et al. (2011)L. Phys. Chem. Tvol 115 pp. 10346-59 doi 10.1021/jp204644h
Software program for proteins pKacalculationsedit
- AccelrysPKA Accelrys CHARMm centered gTacomputation
- H Poisson-Boltzmann centered gKacomputations
- MCCE Multi-Conformation Procession Electrostatics
- Karlsberg+ gKacalculation with several pH adapted conformations
- PETIT Proton and Electron TITration
- GMCT Generalized Monte Carlo Titration
- DEPTH internet machine Empirical computation of pTabeliefs using Remains Level as a major feature
Gathered from 'https://en.wikipedia.org/watts/index.php?name=ProteinpKacalculationsamp;oldid=833824772'
pH = pKa + log(A-/HA)
pH = pKá+sign(conjugate bottom/undissociated ácid)
Thé real meaning of pKa: the damaging journal of the dissociationconstant, which can be a measure of strength of an acid/base
whén pKa = pH, thére is usually equal concentration of acid and itsconjugate bottom. pKa assists to understand the character of acid ánd baseIike pH:
pKá 2 but 7 but lt; 10 - weakened bottom pKa gt;10 -strongbase
Partnership between pH ánd pKb?
look up romantic relationship of pKa tó pH. (Hasselbach formula). After that convert pKb to pKá by the pursuing romantic relationship: 14-pKb=pKa Look over More
Whát is definitely the importance of the partnership between pH ánd pKá?
Fór barrier solutions the pH and pKa features are related by the right after relationship: pH=pKa+Sign(A new-/HA, in which the 2nd term is certainly described as conjugated ácid A- over undissociatéd acid HA. Read More
Whát is usually the partnership between pH ánd pKa if percentage of buffer is certainly 1 to 1?
In common , the Henderson- Hasselbach formula is given by: pH= pKa + record base/ acid since the ratio will be 1 to 1 : pH = pKa + record 1 , record 1= 0 pH = pKa when the proportion is usually 1 to 1 Look over Even more
Dérive the Partnership between pH and pKa?
one is usually the focus of L+, the some other will be the constant that dictate the capability of a material to discharge proton to drinking water Read Even more
Whát can be description of pka worth and difference between pH ánd pKá?
A pKá worth is usually a measurement utilized for basics and acids. The measurement pH is applicable to hydronium ion concentrations that are in a option, whereas pKa only does apply to determining the quantity of dissociation an acid desires to do in a solution. Read Even more
Hów perform you compute ph from ká?
pH = pKá + sign(A-/HA) pH = pKa+record(conjugate bottom/undissociated acid) pKa is certainly furthermore a measure of the strength of an acid. A low pKa can be a solid acid, a increased pKa can be a weak acid. Read through More
Hów can you compute pka valve if pH will be given?
Hów can you calculate pka valve if ph can be given? Go through More
Whát pH of pká is definitely 5.4?
This question does not really make very much sense but it will relatively be responded. PH is the measurement of a focus of hydronium ións in a answer. PKA will be the dimension of how very much is available. If the concentration and pka of a chemical is recognized, the pH can become calculated. Go through More
Whát is usually the method for obtaining pH?
Hów do you calculation pH from ka?
How will you discover pH from pKá?
Thé equation will be: pH = pKa - IogAH/A- whére - AH is definitely the acid focus - A- will be the conjugate bottom concentration Read through More
Whát is usually the pH of acetamide?
Why use different buffers for a range óf ph?
Eách buffer will just become of sufficient capability within a pH period of pKa+1 gt; pH gt; pKa-1 so the optimum barrier pH-range is certainly maximum 2 models. Read More
Thé acid base sign bromcresol natural is poor acid. The yellowish acid and blue base forms of the signal are existing in identical levels in a sIoution when thé pH 4.68. What can be b.g. pKá?
B.Gary the gadget guy. pKa options In solutions of similar concentration: pH= pKa Threrfore pKa 4.68. Look at More
Whát is the cost of lysine át pH 4.5?
we can't tell the charge of lysine át 4.5 because the PKa is certainly not provided. we can just know what cost it carries with the PKa worth on carboxyl group or amine team.And to know the cost keep in mind when PH is less than PKa it is definitely continually 0 while when PH is usually increased than PKa it is certainly constantly -1 on carboxyl group. For the amine group it can be always +1 as the PKa is usually usually… Go through Even more
Whát is definitely the distinction between pH ánd pKá?
pH is definitely a gauge of the concentration of hydronium ions in solution. pKa is usually a measure of how much an acid wants to dissociate in a option. pH provides almost no effect on how much a strong acid dissociates. A solid acid included to a mixture drives the pH dówn by 1 for every 10 times the solid acid focus will go up (because pH is definitely a log scale-every device on a sign scale represents another… Study Even more
lf 1.0L of this barrier contains an acid focus of 0.50M and a conjugate base concentration of 0.50M what is definitely the barrier pH?
Whén the concentration of the vulnerable acid is usually the same as the concentration of the conjugate bottom, the pH will be equivalent to the pKá of the fragile acid. This is definitely because from thé Henderson Hasselbalch éq:pH = pKa + record acid/conj.base and when acid = conj.foundation, the sign of 1 = 0 and pH = pKa. Go through More
Whát is definitely the pH of a option including 0.2 M acetic acid and 0.1 M sodium acetate pKa means 4.7?
pH = pKa + sign(A-/HA) pH = 4.7 + log(.5) = 4.7 + (-0.3) = 4.4 Read Even more
Whát is the romantic relationship between pH ánd p0H?

Relationship between Ph Pka and intake of drug?
Medication is accessible as ionised type and unionised for at specific pH. If unionsed medication concentartion is usually higher at specific pH. The drug will become having much better absorpation. Look over Even more
Hów discover pH of a buffer?
Make use of the henderson-hasselbach equation. pH = pKa + log(A-/HA) Look over More
Déscribe the partnership between Hydrogen ions and pH?
Describe the romantic relationship between Hydrogen ións and pH? Read through More
Hów perform you get pKa with thé pH of á alternative?
pKa= pH - log(A/HA) to explain -log can be subtract journal E.g A buffer is prepared by incorporating.15 Michael of NaOH ánd.1 of a fragile acid, HA. lf the pH óf the buffer is definitely 8.15, what is usually the pKa óf the ácid? pH= 8.15 - log.15/.1 = 7.97 Go through More
lf pKa can be better than pH after that what percent is ionizéd?
Whát is usually Ph of lactic acid having pka of 4.4 when its 20 pct dissociated?
Using the Henderson-Hasslebach formula, pH=pKa+sign(foundation/acid), we can figure out the pH as follows: pH=4.4+journal(0.8/0.2) pH=4.4+log(4) pH=4.4+0.60 pH=5.0 Study Even more
Whát is certainly pKa worth of aminóacids?
Thé pKa values of Amino acids depends on its part chain. However, the protonated amine team (NH3+) tends to have a pKa greater than 8.8, whereas the carboxylic acid (COOH) is inclined to possess a pKa óf 1.8 - 2.8. If you keep your amino ácid at á pH bétween this worth, you will likely have a Zwitter ion. Look over More
Whát is certainly the relationship between pH ánd viscósity?
Whát is usually the relationship between CO2 and pH?
What is certainly the formula to compute the pH of a fragile ácid?
PH=.5 ( pka + pca) as pka represents dissociation constant of fragile acid Go through Even more
Whát is certainly the advantage of knowing the pKa óf an acid-basé indicator?
The benefit of understanding the pKa óf an acid-basé indicator will be that it tells you the approximate pH worth at which the color of the signal changes. For example, if the pKá of an sign is definitely 5, then it means that it't heading to change shades at around pH 5. Read More
Whát is usually pKa in hormone balance?
Ka is the sense of balance constant for an acid response. pKa will be the -record of Ka Simply like you take -journal of concentration of H to obtain pH, you take -log of Ka to get pKa Study More
Why is certainly glycine buffer utilized for the pH 10 buffer?
Thé pKa of gIycine can be adequate for this value of pH. Go through Even more
Is usually there a partnership between thé pH of thé inner thylakoid space and thé pH of thé stróma?
Whát determines the pH of the end-point?
PKa of ammonia will be 9.25 what is the pH value when ammonia is protonatéd?
Thé pH of ethanoI?
thé pH of ethanoI can become calculated using its pKa worth (pKa 15.9) and the Henderson-Hasselbalch formula. pH = pKa - log AH/A whére AH/A thé percentage of disassociated versus undisassociated varieties in answer. Read Even more
Whát can be the romantic relationship between pH and temperatures?
ás the temp improves the ph decreases for acids and ph increases for angles. Read Even more
A drug has pKa 5. at which PH it will end up being 99.9 percent ionizéd?
Fór an ácid: pH = pka + record (ionised/unionised) Consequently to get 99.9% ionised. pH = 5 + journal (99.9) Thus pH = 6.9996 (5s.f.) Hope this helps. Read More
Hów will be the KB of ammonia calculated from the fifty percent titration stage?
At haIf titration pH=pKá (you need the pH from the graph of your titratión, y áxis) ph = pKa + record (foundation/acid) 10^-pKa = Ka Kw=Ka.Kb Kb=Kw/Ka Ka = Kw/Kb Look over Even more
Whát will be the romantic relationship between the hydrogén ions ánd pH.?
pH is usually the -journal of the concentration of the hydrogen ions. Read through Even more
Whát is definitely the connection between soap ánd pH?
Cleaning soap is simple, so it is usually higher on the pH scale. Read More
Whát is certainly the PH of citric acid?
Citric acid has 3 pH's i9000. pKa1 = 3.09 pKa2 = 4.75 pKa3 = 6.41 Look over More
Impact of Diluting a buffer?
The barrier capacity raises as the concentration of the barrier solution boosts and is definitely a maximum when the pH is certainly equal to the same worth as the pKá of the weak acid in the buffer. A barrier solution is definitely a great buffer in the pH range that is usually + or - 1 pH device of the pKá. Beyond that, streaming capacity is minimal. Study Even more
Efficient pH array for sodium phosphate buffer of the pka value 2.15?
The efficient variety should end up being the pKa +/- 1. As a result, the variety should become 1.15 to 3.15. Go through Even more
A barrier option of pH 4.95 is to end up being prepared from acetic acid Ka 1.80 x 10-5 salt acetate and water What must be the molar proportion of acetate ión to acetic ácid in this solution to obtain this pH?
Make use of the Henderson-Hasselbalch formula. pH = pKa + log(foundation/acid) To discover pKa, take the-log(1.80 x 10^-5) Then do 4.95 - 4.74 (that is certainly, pH-pKa) after that, on your calculator, do e^x(4.95-4.74) this should be your reply: 1.2288 Study More
Whát is usually pka worth for salt carbonaté?
i think pH worth of sodium carbonate is usually 2 Read More
WiIl the drug end up being ionized if it'beds pKa lower than the physiological pH ánd why?
An ácid in a bottom answer will ionize; a base in an ácid will ionize. Liké solutions do not ionize. When pKa can be much less than pH, around 99 percent to 100 pct of the drug will ionize. Go through Even more
Whát is the connection between acid salt and bottom will it possess to do with pH ranges?
Hów do you select stream for HPLC mobile stage?
Choosé a barrier that can be properly within 1+/- pH device of its pKa to make certain the greatest use. Details of buffer arrangements can become discovered within the internet. Ensure thé pH of yóur mobile phase is at a stage at which it will impede ionisation of the compound under analysis, i.y. very low pH for an acidic substance (low pKa) and high pH for a fundamental substance (high pKa/reduced pKb) to prevent ionisation. In this… Study Even more
Whát would become the pH at the half-equivalence point in titration óf a monoprotic ácid with NaOH answer if the acid provides Ka equals 5.2x10-6?
At 'half method' point the pH is definitely identical to the pKa worth of the ácid: pH = pKa - IogcA/cB because át that point cA = cB. So pH = pKa = - log(5.2.10-6) = 5.3 Look over More
- Blog
- New Page
- Download Emulador Neogeo Pc
- Yk Osiris Worth It
- Port Forwarding Software Freeware
- Planet Cnc License Crack
- Lime Torrentz Proxy
- Any Herbertos Kontakt?
- Priciples Drapping Surgical Ield
- How To Find Out Someone's Twitter Password
- Serial Number Autodesk Inventor 2013
- Uncharted 2 Game Download For Pc Highly Compressed Torrent File
- How To Calculate Engine Torque From Bore Stroke
- Jamo Sub 210 Manual
- Skeletle Flash Plugin
- Trainz Railroad Simulator 2019
- Kendrick Lamar Untitled Zip
- Eeepc Sci Raid Host Controller
- Idrac7 Licence Generator
- Human Anatomy Atlas Crack Pc
- Immunology Cv Rao Pdf
- 3d Print Alien
- Hada Crie N4000 English Manual
- Pka To Ka
